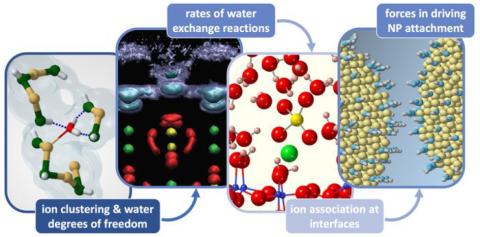
Our understanding of the process of mineral nucleation is starting to evolve from the solely thermodynamical aspects in classical nucleation theory toward a mechanistic study of reactions influenced by solvation structures of ions and their dynamics. A knowledge of how these atomic and molecular interactions respond to changing solution composition and/or the nature of an interface will form a fundamental foundation to understand rate-limiting reactions and processes for nucleation and growth. Here, we review recent advances from multidisciplinary experimental and computational approaches of the structure and dynamics of aqueous solutions and mineral–liquid interfaces that will influence rates of nucleation (and growth behavior) both homogeneously in aqueous solutions and heterogeneously at mineral–liquid interfaces. These processes include structure (and extent) of solute clustering, solvent degrees of freedom, and the presence of noninnocent charge-balancing ions, all of which can drive the selectivity and exert control over nucleation mechanisms. Similar controlling reactions can also occur at interfaces with the added complexities of how solute ions interact with and assemble on surface sites and use different mechanisms from bulk solutes, such as lateral solvent exchanges between distinct surface sites. To gain a predictive understanding of nucleation chemistry, we emphasize several key knowledge gaps that will need to be addressed in this field and highlight the importance of combining computations and experiments.
Publication Solution and Interface Structure and Dynamics in Geochemistry: Gateway to Link Elementary Processes to Mineral Nucleation and Growth
Publication Image
Description
English
