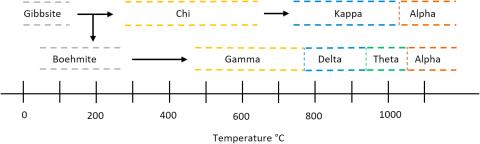
Aluminum hydroxide (gibbsite, Al(OH)3) powders and their thermally dehydrated forms were rehydrated and irradiated with gamma rays to examine the effects of the collapse of the crystalline gibbsite structure as it is converted to alumina on the transport of precursors of molecular hydrogen to the surface. The amount of hydrogen gas, H2, production from irradiated gibbsite and its transition phases with adsorbed water increased substantially with increasing temperature for dehydration of the samples, up to a point. The production of H2 for samples without water was considerably lower than the hydrated samples signifying the importance of surface water and the transport of precursors to the surface. EPR spectroscopy showed that the major radiolytic products are trapped electrons and related O− centers. Thermal gravimetric analysis (TGA) measurements of gibbsite dehydration established the temperatures of phase transitions from gibbsite to alumina. Changes to the surface and structure of gibbsite and its transition phases following irradiation were analyzed using nitrogen adsorption, powder X-ray diffraction (pXRD), Raman spectroscopy, electron paramagnetic resonance (EPR), and scanning electron microscopy (SEM). pXRD and Raman spectra showed amorphorization at about 300 °C, coupled to a substantial increase in specific surface area due to increasing porosity.
Publication - Journal Article Radiolysis of Thermally Dehydrated Gibbsite
Publication Image
Description
English
