Description
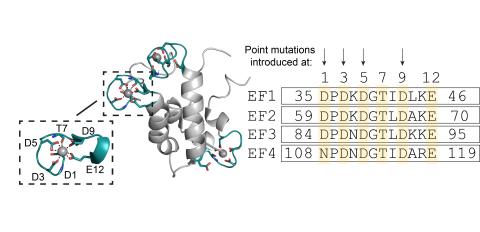
Rare earth elements (REEs) are essential to modern technologies, yet their high physical and chemical similarity makes separation of individual REEs difficult and environmentally taxing. Metalloproteins offer a promising alternative for selective REE binding, as they tend to have high metal ion affinity and specificity. Lanmodulin (LanM), in particular, has arisen as a potential candidate for REE separation as it exhibits picomolar affinity for elements in the REE family. Prior work has shown that the single point mutation D9N can shift LanM’s preference away from lanthanides toward actinides, motivating efforts to tune selectivity of LanM through targeted mutagenesis. Here, we tested the hypothesis that introducing selective aspartic acid to glutamic acid substitutions in the metal coordinating EF hands of LanM would impose steric constraints that would drive LanM affinity away from larger ions, such as La3+, to smaller ions, such as Y3+. To test this hypothesis, a combination of computational and experimental approaches were employed to evaluate the signal mutations LanM D5E and LanM D3E and the double mutants LanM D1ED5E and LanM D3ED9E. Surprisingly, increasing the number of mutations within the metal center did not enhance affinity for smaller REEs, or decrease affinity for larger ions. Only the single point mutation LanM D5E weakened La3+ binding by one order of magnitude relative to LanM wild type (WT), and pairing it with a second mutation to produce LanM D1ED5E drove La3+ affinity to be stronger than that seen for LanM WT. The D3E mutation alone prevented proper expression and folding, but paring it with D9E to produce LanM D3ED9E rescued expression and yielded La3+ affinities comparable to LanM WT. All variants that expressed (LanM D5E, LanM D1ED5E, LanM D3ED9E) displayed Y3+ affinities comparable to LanM WT. Overall, these results highlight the tunability of LanM’s metal-binding environment but also expose current limitations in predicting structural responses to point mutations within a protein sequence. This work establishes a foundation that can be used for refining computational and experimental strategies to engineer metalloproteins with tailored REE selectivity.